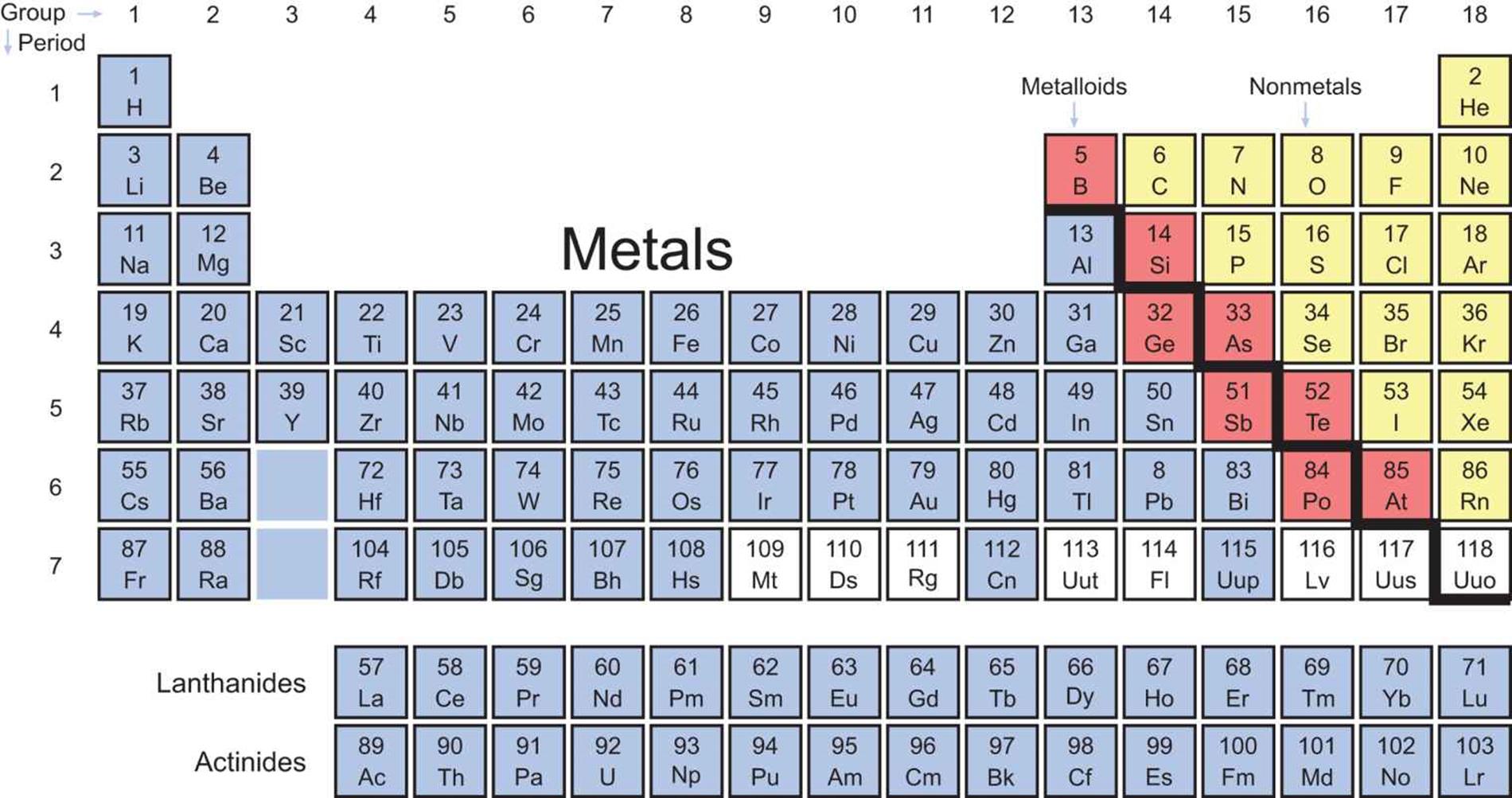 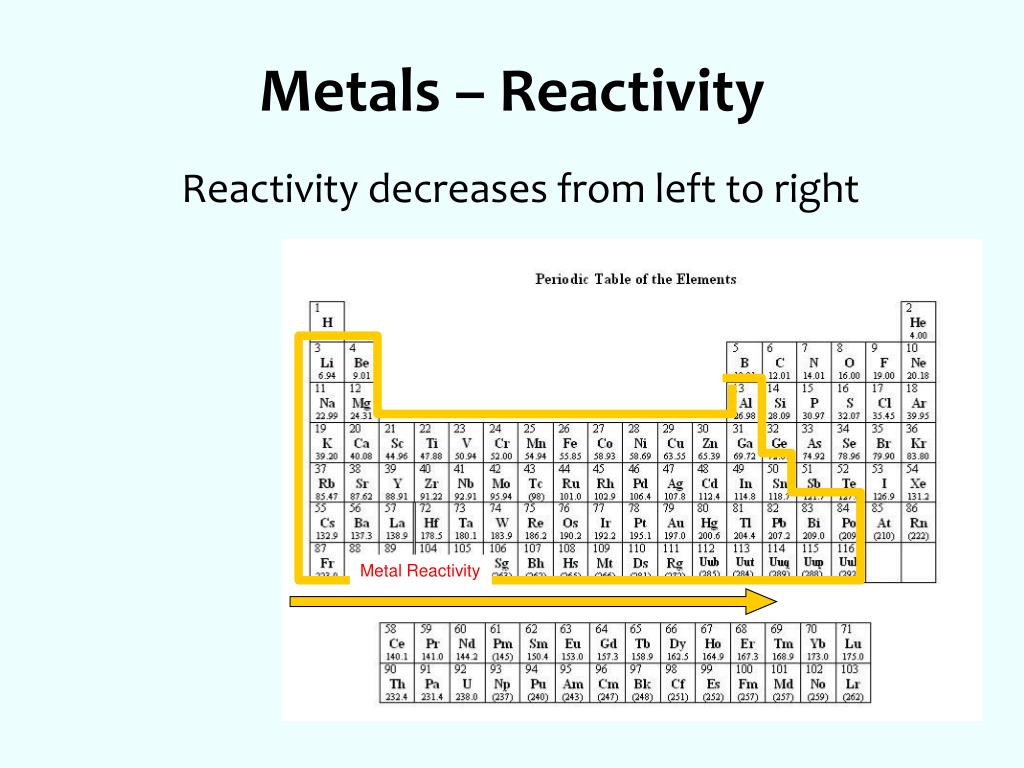
Now compare the reactivity of calcium and magnesium with water. Watch the videos above and compare the reactivity of sodium and potassium with water. View all 118 elements in one interactive Periodic Table. This is a collection of free printable periodic tables in PDF file or PNG image format to save, print, and use. Explore the Periodic Table of Elements and their states, melting points, and boiling points. You can place it where you need it while solving problems, mark it up, and print a new one whenever you like. Notice how reactivity of metals increases as we go down a group. The periodic table lists the elements in order of increasing atomic number and includes other key facts, like atomic weight. Potassium reacts faster than sodium to produce hydrogen gas.Įlements in group 2 also react with water to form hydrogen gas but are not as reactive as group 1 elements. The organization of elements on the periodic table allows for predictions concerning reactivity. Sodium reacts slowly with water to produce hydrogen gas. The periodic table can be used to predict the reactivity of different elements and their behavior in replacement, or dispalcement. It is easier to remove the valance electrons. Weak attraction results in lower ionization energies. Click on the blue writing below to see the reactions. Therefore, the attraction between the nucleus and valance electrons is weak. 
These metals react with water to produce hydrogen gas. These metals react readily with other compounds and elements to give away some of their outer shell (valence) electrons. AllĮlements in group 1 are very reactive metals, with the exception of hydrogen Elements in the same group display similar chemical properties and trends emerge as we go along a period and down a group.Īll elements in a particular group share similar chemical properties. The top metals are more reactive than the metals on the bottom. The table is divided into groups(columns) and periods(rows). The activity series is a chart of metals listed in order of declining relative reactivity. The modern periodic table has elements arranged in order of electronicĬonfiguration. Recognise that elements can be grouped together according to atomic mass. Mendelev was a Russian scientist who was first to Interactive Periodic table Labeled periodic tables. The first example occurs in the case of the lanthanoids (elements having atomic numbers between 57 and 71).The lanthanoids have the general electron configuration Kr4d 10 4f i 5s 2 5p 6 5d 0 or 1 6s 2. Protons, neutrons and electrons of all elements are mentioned in the table below (You will get the List + Shell diagram of all the elements.) Skip to content. The periodic table is a way of organizing known elements into groups with The third major category of elements arises when the distinguishing electron occupies an f subshell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
